National Medical Group (IRENE) in Arab Health 2017
| Welcome to visit us at Arab Health 2017 |
| Naton Medical Group will attend Arab Health 2017.
Welcome to visit us at S1A59!
|
Unique Double Award for Ulrich Medical
In an exceptional collaborative project, a team of trainees from ulrich medical and students from the South Württemberg Student Research Center developed an innovative solution to reduce wear of the milling cutter during machining.
Following initial difficulties, the breakthrough was achieved using a special measurement set-up developed by the project team. "It is extremely rare for patentable technology to result from collaboration of this kind. Yet in this case, we were fortunate and we have submitted it for a patent," Klaus Kiesel, CEO of ulrich medical, was pleased to report. "We are convinced that with the newly developed method, we can optimize not only our own production processes but also other industries can benefit from it."
The new real-time control reduces tool wear by more than 80 percent and decreases consumption of the expensive raw material hard metal which is very energy-intensive to produce. In addition, there is a reduction in manufacturing time which can vary per implant, depending on wear of the milling cutter.
It was not just the jury of the 2016 "Jugend forscht" (Young Researchers) state competition who was impressed by these results. The young researchers were able to reach second place straightaway in the working world specialist field with their project.
ulrich medical was now honored as one of the "100 Companies for Resource Efficiency" and "100 Sites for Industry 4.0" in two more competitions. Both programs were initiated by the state government of Baden-Württemberg.
The initiative "100 Companies for Resource Efficiency" was launched as part of the state strategy for resource efficiency by the Ministry of the Environment of Baden-Württemberg and industry representatives. In addition to energy efficiency, the objective with regard to the topic of resource efficiency in the industry was to once again increase the focus on material efficiency in particular. The collaborative project achieved both objectives. Environment minister Franz Untersteller presented the award in October in Karlsruhe.
With the competition “100 Sites for Industry 4.0 in Baden-Württemberg,” the state of Baden-Württemberg honored innovative concepts from the economy which were successful at intelligently networking production and value-creation processes. In addition to the degree of innovation, the jury of experts also assessed the specific practical relevance for industry 4.0. On November 7, the prize was awarded by the minister of economic affairs Nicole Hoffmeister-Kraut in Stuttgart.
"We are proud to be the only company with its project honored in both future-minded initiatives," says Christoph Ulrich, managing partner of ulrich medical. "Little by little, the procedure developed is now being implemented in all of our production machines. In the medium term, the system is intended to be further developed in such a way that it can be used by all machining centers as well as by other industries."
Fascination with medical technology
ulrich medical develops, produces and markets innovative medical technology. Physicians and users worldwide trust our more than 100 years of experience. With product divisions in spinal implants, contrast media injectors, foot surgery as well as surgical instruments and tourniquets, ulrich medical offers a comprehensive portfolio of innovative products. As one of the few in the industry with development and production in Germany, the medical technology company from Ulm places great value on "Made in Germany".
Immedicom in EFORT Congress 2016

Event : EFORT congress 2016
Period : June 1st(Wed) ~ 3rd(Fri) 2016
Location : PALEXPO, Switzerland
EFFORT congress 2016 was held from June 1st ~ 3rd at PALEXPO. This year is the 167h EPORT congress. About 7,000 person attend this congress every year and it is also held in different European areas every year. We introduced our new product Battery powered system, Video guided catheter, kyphoplasty system and orthopedic medical products and had meeting with many distributors. IMEDICOM CO., LTD. will try to do best to enter the world market by attending main congress and exhibitions.
Cantel Medical Expands its Direct Sales Operations in Canada through the Acquisition of its Endoscopy Assets from Vantage Endoscopy
LITTLE FALLS, N.J., Sept. 26, 2016 /PRNewswire/ -- CANTEL MEDICAL CORP. (NYSE: CMN), a leading global company dedicated to delivering innovative infection prevention products and services, today announced that the Company has expanded its direct sales operations in Canada by acquiring endoscope reprocessing business assets from its Canadian distributor, Vantage Endoscopy, Inc., a subsidiary of Diploma Canada Holdings Ltd. The endoscope reprocessing business consists of Cantel's Medivators®-branded automated endoscope reprocessors, chemistries, consumables and a full sales and service organization.
Jorgen B. Hansen, President and CEO of Cantel Medical said, "This acquisition is consistent with Cantel's strategy to expand its products and services into key markets to provide global solutions that meet local needs. We are pleased to be able to offer customers an expanded Medivators®-branded product portfolio while providing the same level of high-quality service and support they have always received. We look forward to continuing these relationships and partnering with them in the future."
"The expansion of our commercial platform and the addition of a direct sales and service organization for our Endoscopy business will further strengthen Cantel's share in the infection prevention market in Canada." stated Michael Spicer, President, Americas Sales, UK, Canada, Latin America and Global Service. "Expanding our presence in the Canadian market will enable us to realize the full potential of our portfolio while also enabling us to offer a broader suite of infection prevention products from all Cantel divisions."
Ex-Duke football player: How I had my heart attack (and what you can learn from it)
Heart disease is the number one cause of death for both men and women in the United States. Claiming approximately 1 million lives per year, heart attacks alone strike someone every 43 seconds. On January 20, 2014, that someone was me.
That day in January began typically enough with a trip to the gym. I have always been physically active – I played football at Duke University – and I know how exercise benefits the body and the mind. What was not typical however, was the incredible pressure I felt in my chest after twenty minutes on the elliptical machine. I tried to ignore it and hoped it would go away, but when the pressure turned to pain, I knew something was wrong.
When I got home from the gym, I called 9-1-1. I felt cold and clammy, and I was certain I was having some sort of heart event. At the hospital, medical professionals confirmed I was having a heart attack, and I was rushed to the catheter lab to clear any blockages in my arteries. The doctors performed a coronary angioplasty to relieve the blockages, restore blood flow and ease the pressure on my heart.
For me, heart disease is very personal; my father was a heart patient, and – like father, like son – now I was as well. Statistically, that is not that surprising. Black Americans are at a greater risk for cardiovascular disease than white Americans, and black men between the ages of 45 and 64 have a 70 percent higher risk of developing heart failure than white men.
On the way to the catheter lab, lying helpless on the hospital bed, I thought about my father who had quadruple heart bypass surgery years ago.
On the way to the catheter lab, lying helpless on the hospital bed, I thought about my father who had quadruple heart bypass surgery years ago. His surgery was successful, but the recovery period was difficult and lengthy, and it put great a strain on him and on our family. I feared this same scenario would come to pass for me and my family too. Happily for me that was not the case. After only two days in the hospital I returned home feeling better than I had in months.
Heart attack treatment has come a long way. According to the Society for Cardiovascular Angiography and Interventions, a patient who suffered a heart attack in the 1950s was typically treated with weeks of bed rest and pain medications. Survival rates were low. Now, approximately 96 of every 100 heart attack patients who receive treatment not only survive, but are usually released from the hospital and back to work within a week.
A number of innovations have made this dramatic progress possible. Angioplasty is one of these key innovations. In my father’s case, open-heart bypass surgery (CABG) was the only way doctors could restore blood flow to the heart. But advances in technology have led to less invasive approaches. In my case, only a small incision was required to insert the catheter that opened the artery with a balloon-like mechanism and insert the stent to hold the artery open.
Innovative medical technology not only saved my life; it was also the key factor contributing to my speedy recovery. My father was my hero and someone I tried to emulate in many ways, but in this case, it meant I did not have to follow in my father’s footsteps.
Director of Global Marketing Solutions & Support at Marriott International, and a former stand-out Duke football player. He currently resides in Gaithersburg, Maryland, with his wife and two children
Cohera Medical, Inc.® Receives FDA Approval to Begin Clinical Trials of its Sylys® Surgical Sealant in the U.S.
Breakthrough Surgical Sealant Designed to Help Reduce Anastomotic Leaks for Patients Undergoing Colorectal Surgery
RALEIGH, N.C., July 12, 2016 (GLOBE NEWSWIRE) -- Cohera Medical, Inc., a leading innovator and developer of absorbable surgical adhesives and sealants, today announced that it has received Investigational Device Exemption (IDE) approval from the United States Food and Drug Administration (FDA) to begin a prospective, multicenter, randomized clinical trial for its Sylys® Surgical Sealant device.
/EIN News/ -- Sylys® Surgical Sealant is intended to be used as an adjunct to standard closure techniques for the reinforcement and protection of anastomotic junctions in colorectal and ileorectal anastomosis procedures. Sylys is a resorbable synthetic sealant designed to help reduce anastomotic leakage by providing additional support to the anastomosis during the first few days of healing, when the development of leaks is most likely to occur.
“It is tremendous that the FDA has given investigational approval to this novel device that may help reduce the incidence of anastomotic leaks and their devastating impact,” said Dr. Deborah Nagle, Chief, Colon and Rectal Surgery, Beth Israel Deaconess Medical Center in Boston, MA. Sylys® Surgical Sealant received Expedited Access Pathway (EAP) status from the FDA in 2015, which recognizes the potential of the product to meet a serious unmet clinical need. This IDE approval represents the culmination of significant joint efforts by the company and the FDA to move forward on the clinical evaluation of this important technology.
“Anastomotic leakage is the most devastating complication associated with intestinal resection, contributing to morbidity and mortality,” said James McCormick, DO, FACS, FASCRS, Program Director, General Surgery, Allegheny Health Network in Pittsburgh, PA. “We have made tremendous strides in curtailing the risk associated with intestinal anastomosis, but we are always striving for further improvement and greater patient safety.”
The randomized clinical study will compare patients undergoing colorectal and ileorectal anastomosis after resection with and without the sealant. The study is the first phase of a clinical trial program designed to support the safety and efficacy of the sealant in reducing the leak rate in these procedures.
Anastomotic leakage, which occurs in up to 23 percent of patients undergoing colorectal surgery, is considered to be the most serious surgical complication encountered, frequently resulting in the rapid development of severe peritonitis, septic shock, multiple organ dysfunction, and death. At least one third of post-surgical deaths following colorectal surgery are attributed to leaks, and survivors generally have protracted and expensive recoveries.
“We are pleased that the FDA has approved this IDE. This approval validates the Sylys® Surgical Sealant biocompatibility, pre-clinical safety profile, and clinical trial design of the study,” said Chad Coberly, JD, Chief Clinical and Legal Officer of Cohera. “The Company greatly appreciates the cooperation and high level of interaction with the FDA during the review of this potentially life-saving healthcare innovation.”
The market for Sylys® Surgical Sealant is significant, with more than one million procedures per year worldwide representing a multi-billion dollar opportunity. Due to the unmet clinical need, Sylys would represent a breakthrough in this market that will lead to improved patient outcomes as well as reduced patient management costs for healthcare providers.
Cantel Medical Names Jorgen B. Hansen as New President and CEO
LITTLE FALLS, N.J., Aug. 1, 2016 /PRNewswire/ -- CANTEL MEDICAL CORP. (NYSE: CMN) announced today the appointment of Jorgen B. Hansen as the Company's President and Chief Executive Officer, effective today, consistent with the Company's long-term CEO succession plan. Mr. Hansen also will become a member of the Board of Directors. Mr. Hansen succeeds Andrew A. Krakauer, who will serve as a Senior Advisor for the Company until October 15, 2016.
Charles M. Diker, Cantel's Chairman of the Board, stated, "We brought Jorgen Hansen to Cantel nearly four years ago. He's a dynamic, experienced leader in the medical device industry who has clearly demonstrated strong leadership and the vision necessary to achieve our full potential. In addition to leading the day-to-day operations for the Company, Jorgen played a key role in the development of our strategic plan including our expansion in international markets and the successful completion of 12 acquisitions. All of these accomplishments have reinforced our confidence in Jorgen's ability to lead the Company as Chief Executive Officer."
Mr. Diker added "I also would like to thank Andy Krakauer for his exceptional service to the Company over the past 12 years. He has been an outstanding leader who helped Cantel achieve exceptional success during his tenure. His contributions and experience in the medical device industry have been invaluable to the Company."
Mr. Hansen joined Cantel in November 2012 as EVP and COO, and was appointed to President and COO in November 2014. He also served as President and CEO of Cantel's Endoscopy business unit, the Company's largest division, from November 2012 to July 2015. Prior to joining Cantel, Mr. Hansen held leadership positions with increasing responsibility in the global medical device industry. He worked at ConvaTec from May 2009 - November 2012, most recently as Senior Vice President, Global Marketing, Business Development, Science and Innovation. Mr. Hansen joined ConvaTec from Coloplast, where he held various leadership roles over a 13-year period in Asia and Europe, ranging from General Manager, Division Head and Senior Vice President of Global Operations. He received a Bachelor of Science Degree in Mechanical Engineering from Denmark Technical University, and an International Commerce degree from the Copenhagen Business School in Denmark.
"I am honored to be appointed as Cantel's CEO and lead this exciting company and talented team," said Mr. Hansen. "I would like to thank Mr. Diker and the Board for this opportunity, and as CEO, I will continue to pursue Cantel's vision to be the global leader in infection prevention. We will do this by executing on our business priorities focused on new products, market expansion and M&A, supported by continuous improvement."
Welch Allyn to add 100 jobs in Skaneateles
Skaneateles, N.Y. — Welch Allyn plans to add 100 new jobs to its medical equipment factory in Skaneateles, already one of Central New York's largest employers.
Company President Alton Shader and Gov. Andrew Cuomo made the jobs announcement today at Welch Allyn's headquarters on State Street Road, where the company employs 900 people.
Cuomo said the state will provide the company with $12 million in incentives. Onondaga County also will provide economic development incentives to the company, he said.
Chicago-based Hill-Rom Holdings Inc. bought the company for $2.05 billion in September from the Allyn family and has continues to manufacture medical diagnostic equipment under the Welch Allyn name in Skaneateles.
The acquisition raised fears in the community that Hill-Rom might pull jobs out of Skaneateles. Two days after the acquisition, Hill-Rom laid off 50 people at the plant, but there have been no major layoffs since then, and today's announce came as welcome news.
Shader said Welch Allyn has had the most successful year in its history.
"The future is bright for Welch Allyn," he said.
He said the jobs will be in manufacturing, marketing, and research and development and will be created over the next two years. The state's economic development incentives are contingent on the company creating the jobs, he said.
Sen. Charles Schumer, D-NY, did not attend the announcement in Skaneateles but issued a statement saying that a recent delay in implementation of a special federal tax on medical equipment sales played a role in the company's decision to add the jobs. Schumer had pushed for the delay.
"Simply put, today's announcement is proof that the medical device tax needs to be permanently repealed so Welch Allyn will remain a source of economic energy for Central New York for years to come," he said.
Dr. Francis Welch and William Noah Allyn founded the company in Skaneateles in 1915.
A Visual Guide to Posterolateral Gutter Fusion Surgery
A spinal fusion is a surgical procedure that joins your spinal segments together for the purpose of eliminating motion at that segment. The overall goal of this type of procedure is to decrease your pain.
Watch: Spine Fusion Surgery Video
Our video walk-through can help you better understand the gold standard of spinal fusions, the posterolateral gutter fusion.
Video highlights
A posterolateral gutter fusion is performed on your lower back (lumbar spine) to relieve pain from lumbar degeneration.
See Lumbar Degenerative Disc Disease (DDD)

The image above pictures the spinal segments where a posterolateral gutter fusion is performed: L1-L2 through L5-S1 at the bottom of your spine. The segments that most often require a fusion are at L4-L5 and L5-S1.
Watch: Lumbar Spine Anatomy Video
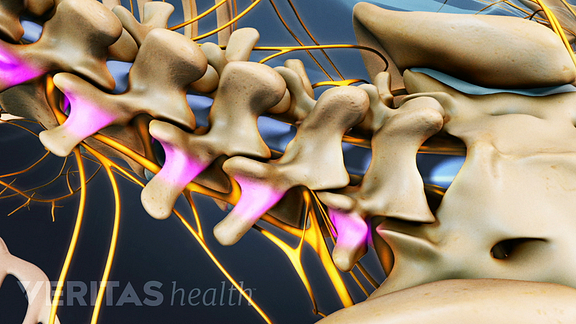
To stop the motion at your affected segment, a bone graft is placed along the posterolateral portion of your back (shown above in purple).
See Autograft: The Patient's Own Bone
The procedure
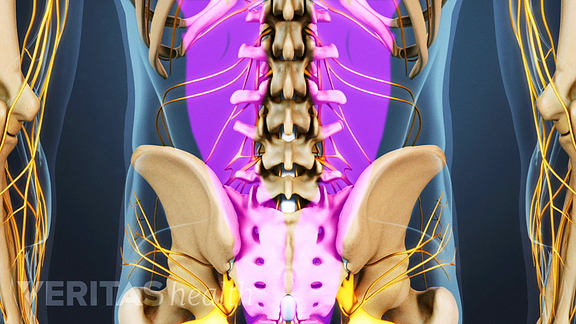
To begin, your surgeon will make a 3 to 6 inch incision in the midline of your back, and then retract the muscles surrounding your spine. In the above image, the muscles are shown in purple; and you can see the exposed spine.
See Back Muscles and Low Back Pain
At this time, your surgeon may perform a laminectomy to remove any bone that is irritating your nerves.
Watch: Lumbar Laminectomy Surgery Video
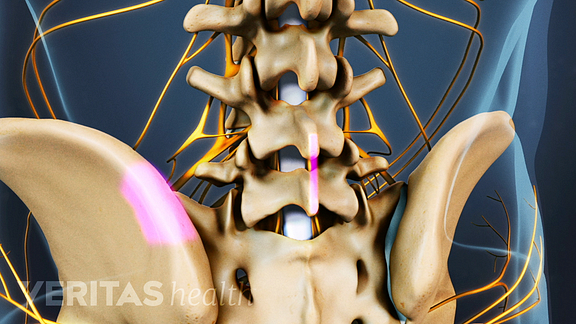
Next, your surgeon will retrieve bone for the graft from your iliac crest, which is the uppermost part of the back of your pelvis. Your surgeon may be able to retrieve the bone graft through the original incision on the midline of your back.
See Synthetic Bone Graft Options
In some cases, a synthetic or cadaver bone graft may also be used.
See Allograft: Cadaver Bone from a Tissue Bank

Once the bone graft is ready, your doctor will expose the posterolateral area of your back by removing the muscles that attach to the transverse processes.
This area of your body has a high number of blood vessels, which are crucial for helping your body generate new tissue for the bone graft. Your surgeon will place the bone graft morsels (shown above as reddish cells) on the exposed posterolateral area.

Your surgeon will then place your muscles back down over your graft. This generates tension that will hold it in place as your body heals.

This process is then repeated on the other side of your spine so that both sides will fuse together.
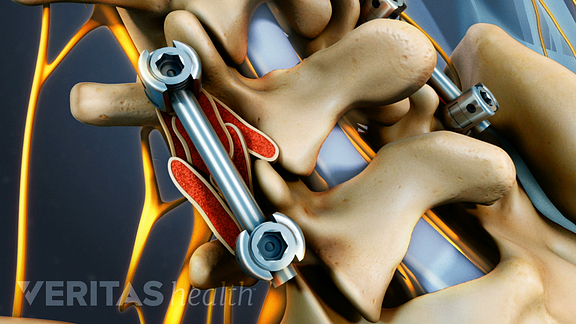
In certain cases, your surgeon may use pedicle screws and rods to provide additional stability for your bone while it is fusing.
See Spine Fusion Instrumentation

Results from a posterolateral gutter fusion
The image above pictures the result of the fusion after several months of healing: The body's natural healing responses have helped the bone graft and adjacent vertebral bodies fuse into a single bone.
See After Posterolateral Gutter Fusion Surgery: Risks and Complications
A successful fusion surgery is partially dependent on several factors, including the skill of your surgeon and leading a reasonably healthy lifestyle.
The main risk with this type of procedure is that a solid fusion will not be obtained.
See Failed Spinal Fusion Surgery
Failure rates are higher for patients who:
- Have had prior surgery
- Smoke or are obese
- Have multiple level spine fusion surgery
- Have been treated with radiation for cancer.
See Quitting Smoking Before a Spinal Fusion
Tiger Woods Undergoes Microdiscectomy Surgery for a Pinched Nerve
Tiger Woods is back.
After a 15 month absence from professional golf, Woods is competing in the Hero World Challenge event this week. This is Woods' first event since September of 2015, when he underwent a second microdisectomy surgery (he also underwent a follow-up procedure a month later).
With microdisectomy surgery back in the news, we thought it would be a good time to explain this relatively common procedure.
Need for a microdisectomy surgery
Before we talk about how the surgery works, lets quickly look at why someone might need a microdisectomy surgery.
A small portion of your bone, or disc material from a herniated disc, can irritate one of your nerve roots. This in turn can result in considerable leg pain. A microdisectomy surgery removes the irritant, and allows space for your nerve to heal.
The procedure
A microdisectomy surgery to remove a disc fragment begins with a roughly 1 inch incision in the midline of your lower back.
Watch: Microdiscectomy Surgery Video: A Spine Surgeon Explains the Procedure
Your surgeon will then perform the following steps:
- Your back muscles are lifted off the bony arch of your spine.
- Next, a membrane over your nerve roo] is removed.
- On occasion, a portion of your inside facet joint is removed to allow access to your nerve root, and to relieve the pressure over your nerve.
- Finally, your nerve root is moved to the side, and the disc material is removed from under your nerve root.
See Spinal Cord and Spinal Nerve Roots
The success rate for a microdisectomy surgery is estimated to be between 90 and 95 percent.
Risks and complications
While rare, there are risks associated with a microdisectomy procedure; including any of the following:
- Nerve root damage
- Bowel/bladder incontinence
- Bleeding
- Infection
See Microdiscectomy Spine Surgery: Risks, Complications, and Success Rates
I hope all of the above information helps you better understand microdisectomy surgery. And we here at Spine-health wish Tiger Woods all the best as he returns to the world of golf following his microdisectomy surgery.












