Cantel Medical Names Jorgen B. Hansen as New President and CEO
LITTLE FALLS, N.J., Aug. 1, 2016 /PRNewswire/ -- CANTEL MEDICAL CORP. (NYSE: CMN) announced today the appointment of Jorgen B. Hansen as the Company's President and Chief Executive Officer, effective today, consistent with the Company's long-term CEO succession plan. Mr. Hansen also will become a member of the Board of Directors. Mr. Hansen succeeds Andrew A. Krakauer, who will serve as a Senior Advisor for the Company until October 15, 2016.
Charles M. Diker, Cantel's Chairman of the Board, stated, "We brought Jorgen Hansen to Cantel nearly four years ago. He's a dynamic, experienced leader in the medical device industry who has clearly demonstrated strong leadership and the vision necessary to achieve our full potential. In addition to leading the day-to-day operations for the Company, Jorgen played a key role in the development of our strategic plan including our expansion in international markets and the successful completion of 12 acquisitions. All of these accomplishments have reinforced our confidence in Jorgen's ability to lead the Company as Chief Executive Officer."
Mr. Diker added "I also would like to thank Andy Krakauer for his exceptional service to the Company over the past 12 years. He has been an outstanding leader who helped Cantel achieve exceptional success during his tenure. His contributions and experience in the medical device industry have been invaluable to the Company."
Mr. Hansen joined Cantel in November 2012 as EVP and COO, and was appointed to President and COO in November 2014. He also served as President and CEO of Cantel's Endoscopy business unit, the Company's largest division, from November 2012 to July 2015. Prior to joining Cantel, Mr. Hansen held leadership positions with increasing responsibility in the global medical device industry. He worked at ConvaTec from May 2009 - November 2012, most recently as Senior Vice President, Global Marketing, Business Development, Science and Innovation. Mr. Hansen joined ConvaTec from Coloplast, where he held various leadership roles over a 13-year period in Asia and Europe, ranging from General Manager, Division Head and Senior Vice President of Global Operations. He received a Bachelor of Science Degree in Mechanical Engineering from Denmark Technical University, and an International Commerce degree from the Copenhagen Business School in Denmark.
"I am honored to be appointed as Cantel's CEO and lead this exciting company and talented team," said Mr. Hansen. "I would like to thank Mr. Diker and the Board for this opportunity, and as CEO, I will continue to pursue Cantel's vision to be the global leader in infection prevention. We will do this by executing on our business priorities focused on new products, market expansion and M&A, supported by continuous improvement."
Welch Allyn to add 100 jobs in Skaneateles
Skaneateles, N.Y. — Welch Allyn plans to add 100 new jobs to its medical equipment factory in Skaneateles, already one of Central New York's largest employers.
Company President Alton Shader and Gov. Andrew Cuomo made the jobs announcement today at Welch Allyn's headquarters on State Street Road, where the company employs 900 people.
Cuomo said the state will provide the company with $12 million in incentives. Onondaga County also will provide economic development incentives to the company, he said.
Chicago-based Hill-Rom Holdings Inc. bought the company for $2.05 billion in September from the Allyn family and has continues to manufacture medical diagnostic equipment under the Welch Allyn name in Skaneateles.
The acquisition raised fears in the community that Hill-Rom might pull jobs out of Skaneateles. Two days after the acquisition, Hill-Rom laid off 50 people at the plant, but there have been no major layoffs since then, and today's announce came as welcome news.
Shader said Welch Allyn has had the most successful year in its history.
"The future is bright for Welch Allyn," he said.
He said the jobs will be in manufacturing, marketing, and research and development and will be created over the next two years. The state's economic development incentives are contingent on the company creating the jobs, he said.
Sen. Charles Schumer, D-NY, did not attend the announcement in Skaneateles but issued a statement saying that a recent delay in implementation of a special federal tax on medical equipment sales played a role in the company's decision to add the jobs. Schumer had pushed for the delay.
"Simply put, today's announcement is proof that the medical device tax needs to be permanently repealed so Welch Allyn will remain a source of economic energy for Central New York for years to come," he said.
Dr. Francis Welch and William Noah Allyn founded the company in Skaneateles in 1915.
A Visual Guide to Posterolateral Gutter Fusion Surgery
A spinal fusion is a surgical procedure that joins your spinal segments together for the purpose of eliminating motion at that segment. The overall goal of this type of procedure is to decrease your pain.
Watch: Spine Fusion Surgery Video
Our video walk-through can help you better understand the gold standard of spinal fusions, the posterolateral gutter fusion.
Video highlights
A posterolateral gutter fusion is performed on your lower back (lumbar spine) to relieve pain from lumbar degeneration.
See Lumbar Degenerative Disc Disease (DDD)

The image above pictures the spinal segments where a posterolateral gutter fusion is performed: L1-L2 through L5-S1 at the bottom of your spine. The segments that most often require a fusion are at L4-L5 and L5-S1.
Watch: Lumbar Spine Anatomy Video
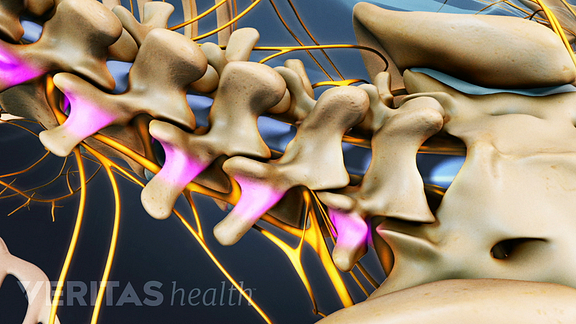
To stop the motion at your affected segment, a bone graft is placed along the posterolateral portion of your back (shown above in purple).
See Autograft: The Patient's Own Bone
The procedure
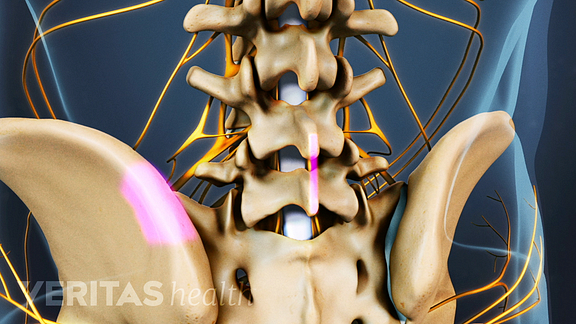
To begin, your surgeon will make a 3 to 6 inch incision in the midline of your back, and then retract the muscles surrounding your spine. In the above image, the muscles are shown in purple; and you can see the exposed spine.
See Back Muscles and Low Back Pain
At this time, your surgeon may perform a laminectomy to remove any bone that is irritating your nerves.
Watch: Lumbar Laminectomy Surgery Video
Next, your surgeon will retrieve bone for the graft from your iliac crest, which is the uppermost part of the back of your pelvis. Your surgeon may be able to retrieve the bone graft through the original incision on the midline of your back.
See Synthetic Bone Graft Options
In some cases, a synthetic or cadaver bone graft may also be used.
See Allograft: Cadaver Bone from a Tissue Bank

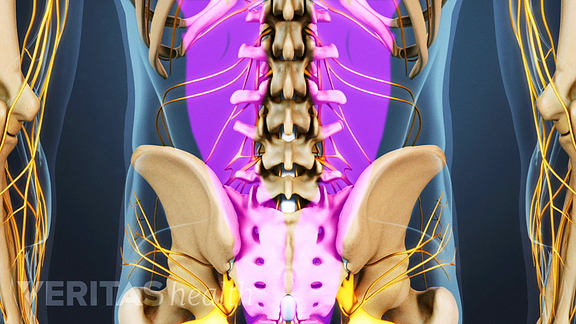
Once the bone graft is ready, your doctor will expose the posterolateral area of your back by removing the muscles that attach to the transverse processes.
This area of your body has a high number of blood vessels, which are crucial for helping your body generate new tissue for the bone graft. Your surgeon will place the bone graft morsels (shown above as reddish cells) on the exposed posterolateral area.

Your surgeon will then place your muscles back down over your graft. This generates tension that will hold it in place as your body heals.

This process is then repeated on the other side of your spine so that both sides will fuse together.
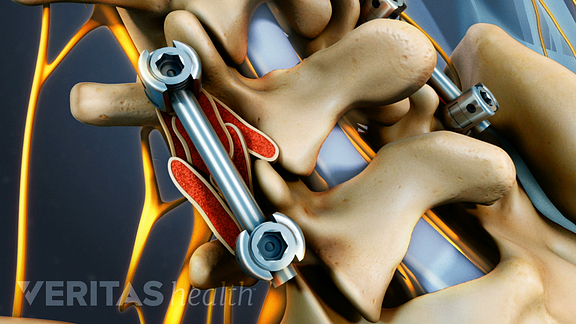
In certain cases, your surgeon may use pedicle screws and rods to provide additional stability for your bone while it is fusing.
See Spine Fusion Instrumentation

Results from a posterolateral gutter fusion
The image above pictures the result of the fusion after several months of healing: The body's natural healing responses have helped the bone graft and adjacent vertebral bodies fuse into a single bone.
See After Posterolateral Gutter Fusion Surgery: Risks and Complications
A successful fusion surgery is partially dependent on several factors, including the skill of your surgeon and leading a reasonably healthy lifestyle.
The main risk with this type of procedure is that a solid fusion will not be obtained.
See Failed Spinal Fusion Surgery
Failure rates are higher for patients who:
- Have had prior surgery
- Smoke or are obese
- Have multiple level spine fusion surgery
- Have been treated with radiation for cancer.
See Quitting Smoking Before a Spinal Fusion
Tiger Woods Undergoes Microdiscectomy Surgery for a Pinched Nerve
Tiger Woods is back.
After a 15 month absence from professional golf, Woods is competing in the Hero World Challenge event this week. This is Woods' first event since September of 2015, when he underwent a second microdisectomy surgery (he also underwent a follow-up procedure a month later).
With microdisectomy surgery back in the news, we thought it would be a good time to explain this relatively common procedure.
Need for a microdisectomy surgery
Before we talk about how the surgery works, lets quickly look at why someone might need a microdisectomy surgery.
A small portion of your bone, or disc material from a herniated disc, can irritate one of your nerve roots. This in turn can result in considerable leg pain. A microdisectomy surgery removes the irritant, and allows space for your nerve to heal.
The procedure
A microdisectomy surgery to remove a disc fragment begins with a roughly 1 inch incision in the midline of your lower back.
Watch: Microdiscectomy Surgery Video: A Spine Surgeon Explains the Procedure
Your surgeon will then perform the following steps:
- Your back muscles are lifted off the bony arch of your spine.
- Next, a membrane over your nerve roo] is removed.
- On occasion, a portion of your inside facet joint is removed to allow access to your nerve root, and to relieve the pressure over your nerve.
- Finally, your nerve root is moved to the side, and the disc material is removed from under your nerve root.
See Spinal Cord and Spinal Nerve Roots
The success rate for a microdisectomy surgery is estimated to be between 90 and 95 percent.
Risks and complications
While rare, there are risks associated with a microdisectomy procedure; including any of the following:
- Nerve root damage
- Bowel/bladder incontinence
- Bleeding
- Infection
See Microdiscectomy Spine Surgery: Risks, Complications, and Success Rates
I hope all of the above information helps you better understand microdisectomy surgery. And we here at Spine-health wish Tiger Woods all the best as he returns to the world of golf following his microdisectomy surgery.
Global Spine Congress 2017 Calendar
EVENTS
- 12.12.2016 - 16.12.2016, AO Davos Courses, Davos, Switzerland
- 12.04.2017 - 14.04.2017, ISASS17, Boca Raton, Florida, USA
- 19.04.2017 - 22.04.2017, Coluna 2017, Rio De Janeiro, Brazil
- 26.04.2017 - 28.04.2017, 6th International Congress on Biotechnologies for Spinal Surgery , Berlin, Germany
- 24.05.2017 - 26.05.2017, 33rd Annual Meeting of the Cervical Spine Research Society – European Section , Salzburg, Austria
- 28.05.2017 - 01.06.2017, 13th ISRS (International Stereotactic Radiosurgery Society) Congress, Montreux, Switzerland
- 11.10.2017 - 13.10.2017, Eurospine, Dublin, Ireland
- 30.11.2017 - 02.12.2017, 38th SICOT Orthopaedic World Congress, Cape Town, South Africa
GS Medical USA Experiences Growth, Expansion and New Product Development in 2015
Irvine (gsmedicalusa.com) – January 19, 2016 – GS Medical USA, LLC, (GSM) worldwide leader in the development and manufacturing of medical devices for the care of spine pathologies, announced today that 2015 represented a landmark shift in both the leadership and direction of the company.
James Shin, CEO of GS America summarized, “2015 was a very successful year for GSM. Not only were we able to add significant resources to our R&D teams in the way of technology, machinery and team members, we were also awarded two new patents and FDA clearance for several innovative products. In addition to our R&D teams, we have made tremendous advancements with talented leadership and in our quality, operations and sales teams.”
“People are the focus of our attention,” explained Brian Oliver, VP of Sales and Marketing, “and we must first concentrate on the well-being of patients and customers. Our priority remains finding solutions to improve the care of patients while delivering impeccable service and technology to our surgeons and distributors. I am very pleased with the progress and growth within GSM during the past year. Our surgeons and distributors continue to speak highly of their interactions and relationships with GSM, and this is only the beginning!” The company is also thrilled to announce the launch of a new website, which will serve as a platform to continue to provide customers with real time information about the growth and development of GSM.
With investments in sales, R&D, engineering and marketing, GSM has laid a solid foundation from which to propel sales and revenue forward in 2016. Through partnerships with local and national GPOs and IDNs, GSM will continue to bring quality, value and cost-conscious solutions to hospitals, surgery centers and patients. “Having the conversation with our surgeons and hospitals about the breadth of treatment options that are available in the care of spine maladies is very rewarding.” states Heather Cain, Clinical Resource Manger. “The greatest reward is when we as a comprehensive patient care team can identify procedural, product and economic solutions to help our customer meet their economic goals.”
The vision for GSM in 2016 is clear: more solutions for new product, value and quality to the surgeon and hospital, and continued growth and expansion via distribution, partnership, and education on the sales front. With the new year well underway, GSM will continue to remain committed to excellence in all aspects of its mission and vision.
About GS Medical USA, LLC
GS Medical USA features many spinal products spanning lumbar, thoracic, and launching new cervical applications. GSM products have been used in thousands of spine surgeries throughout the USA, and hundreds of thousands globally. Staffed with a network of industry leaders, medical sales distributors, engineers, and collaborating physicians, GSM is at the forefront of high-quality, value-added medical technology with a number of innovative implant solutions in the commercialization pipeline. Corporate headquarters are in Irvine, California and with international offices located in Seoul, Korea.
J&J's DePuy to End 2 Hip Replacement System
| Johnson & Johnson's DePuy orthopedics business plans to stop selling a couple of hip replacement systems not widely used by doctors.
DePuy will discontinue both its Ultamet Metal-on-Metal Articulation and Complete Ceramic-on-Metal Acetabular systems worldwide at the end of August. It made the decision as part of a push to streamline its product portfolio. DePuy said Thursday the discontinuations were not connected to concerns about safety or effectiveness, and they were not recalls. J&J, based in New Brunswick, N.J., has been hampered by an embarrassing spate of product recalls of artificial joints, contact lenses, prescription drugs and over-the-counter medicines that began in 2009. DePuy recalled two artificial joint systems — the ASR Hip Resurfacing System and the ASR XL Acetabular System — in 2010 because of unexpectedly high failure rates. DePuy said the Ultamet and Complete brands now represent less than 1 percent of its bearings sold in the United States and Europe. Physicians have come to prefer metal-on-polyethylene, ceramic-on-polyethylene and ceramic-on-ceramic bearings. Polyethylene is a medical-grade form of plastic. The company said it also made its decision due to proposed Food and Drug Administration changes that would require a more extensive approval process for metal-on-metal systems. Ultamet is DePuy's only metal-on-metal system, and the company noted that clinical demand for this type of hip replacement is extremely low. Artificial joint systems replace deteriorated joints causing severe pain and limiting mobility. Hip replacement parts include the top of the thigh bone, the ball atop that and a plate lining the hip socket. The systems are generally expected to last at least 10 to 20 years. DePuy said it will announce other worldwide product discontinuations this year and next, as it simplifies its portfolio. Shares of J&J climbed 19 cents to $87.64 in midday trading, while broader trading indexes also rose less than 1 percent. The stock reached a new, 52-week high price of $88.07 earlier in the morning, and J&J shares have climbed about 25 percent so far this year. |
First "handshake" between Chinese and Russian General Military Hospital
On March 2, 2015, the orthopaedic experts from Russian General Military Hospital visited General Hospital of the People's Liberation Army (PLAGH), the biggest military general hospital in China, and had an in-depth academic change with Chinese experts in Beijing, China.
After visiting the hospital, Chinese and Russian orthopedic experts had a lively cross-border medical seminar in both English and Russian. The two top countries orthopedic experts had a colorful and vivid academic exchange and experience report under orthopedic and trauma emergency topic.
ZIMMER KNEE RECALL
Zimmer Recalls NexGen Knee Replacement Component Due to Risk of Failure - Zimmer NexGen Knee Failure Rate May Be Unusually High - Lawyers Offer Free Zimmer NexGen Knee Case Review.
Prominent joint replacement surgeons, including one of Zimmer's most highly regarded former orthopedic surgeon consultants, have issued stern warnings about the long-term viability of select Zimmer NexGen knee replacement systems. Major publications have reported on multiple medical studies by different orthopedic surgeons for different Zimmer NexGen knee replacement systems that show an unacceptably high number become loose at the femoral component and require revision surgery to be corrected. While most of the Zimmer NexGen knee replacement systems are not experiencing problems, other are - both those recalled & those not recalled.
This website contains information about the Zimmer knee recall and claims you or a loved one may make if you have a Zimmer NexGen knee replacement system that has loosened or is otherwise causing you pain and/or a decrease in mobility.
Wontech Host Symposia W in Seoul
Wontech, a leading innovator of aesthetic and medical advanced laser and related technology, took place a special meeting “Symposia W” at Chrysanthemum Room of Seoul Grand Intercontinental Hotel on 3rd of September, 2016. “Symposia W” is periodical event which host more than 2 times a year, including to place in domestic and international region. In the symposium, there are plenty of remarkable clinical issues with the evidences among reputational dermatologists, and it’s a good meeting to share their own experiences and consultation.
.jpg)
The WonTech’s 'Symposia W' theme is "Beyond the Nano, into the Pico, present and future of the long-pulse laser". Lecture programs are ▲ 'What is Pico laser?, Clinical application of PICOCARE' by Dr. Yeo Euncheol, director of Jongro S & U dermatology clinic ▲ ‘Preclinical studies of PICOCARE’s color tattoo apply’ by Dr. An Jincheol, pre-med chairman of Dankook University ▲ ‘1064nm & 1320nm Mercury Laser experience’ by Dr. Seo Seokbae, director of SeoAhSong dermatology clinic. Dr. Park GiBeon, director of Seoul dermatology clinic was anchored as a facilitator. This seminar was focused clinical studies and application plan of WonTech’s representative equipment PICOCARE.
|
|
WonTech’s representative equipment 'PICOCARE’ is newest device which can be delivers laser energy with 1,000 times shorter than conventional Nanosecond laser that minimize the damage of skin and destroy various pigment at the same time. It approved KFDA last May and getting a lot of attention from domestic and overseas because of better functions and performance to compare with the world's top brands devices. Currently, famous dermatology clinics such as SeoAhSong dermatology clinic, WeedWin dermatology clinics and others use PICOCARE.
A president of Wontech, Mr. Jong won Kim had announced that “Symposia W” was hosted by sharing and discussion of valuable clinical information, and Wontech keep having a plan to host a meeting to the users throughout varied programs.











